
This can allow us to determine some trends in bond lengths. Historically, the Debye was defined in terms of the dipole moment resulting from two equal charges of opposite sign and separated by 1 Ångstrom ( 10 10 m) as 4.801 D from Equation 12.4.7. Please note that this particular table is in picometers (10 -12), and that a common unit for bond length is the Angstrom Å (10 -10), so for example, the O=O bond is 1.207 Å.Ĭoulomb's law states that the bond energy is inversely related to the bond length (r), and so factors which influence a bond's strength influence its length. This is because one has to lose a negatively charged electron and another has to gain one. Table 8.9.1 shows bond lengths for a variety of bonds. This section presents Coulomb’s law and points out its similarities and differences with respect to Newton’s law of universal gravitation. ( 1 vote) Flag Edward 7 years ago When ionic bonds form, one atom becomes positively charged, while the other becomes negatively charged. 
Also, the closer together the charges are, the stronger the attraction. So, for example, Mg 2 + and O 2-will have a stronger attraction than Na + and Cl-, because of the larger charges. Calculate the time using the current and the coulombs of charge. According to Coulomb's Law, the larger the magnitude of the charges on each particle, the stronger the attraction will be. 8.9.1), where the bottom of the well represents the equilibrium position of the oscillating atoms, which we call the bond length. Convert the moles of electrons into coulombs of charge using Faraday's constant. An electrochemical unit of charge, the faraday, is useful in describing electrolysis reactions, such as in metallic. From the definition of the ampere, the electron itself has a negative charge of 1.602176634 × 10 19 coulomb. The idea of a coulomb is useful in electrochemistry and physics. Shares of electric vehicle charging equipment and services company ChargePoint Holdings (NYSE: CHPT) are down 57 so far in 2023, and a brutal 88 from the all-. One coulomb consists of 6.24 × 10 18 natural units of electric charge, such as individual electrons or protons. This article will show you the lattice energy equation, lattice energy formula, how to calculate it step by step, and give you examples of how the equation & formula can be utilized. It is named after Charles-Augustin de Coulomb. NaClsNa+g+Cl-(g) Here, the energy that is provided to 1 mole of NaCl to isolate it into gaseous Na + and Cl ions is 786.0 kJ. 
It is equivalent to 6.241 x 10 18 electrons. The coulomb (symbol: C) is the SI unit of electric charge. The charge may be either positive or negative. This structure suggests that there is a strong attractive Coulomb interaction between nearest-neighbors ions, which is responsible. 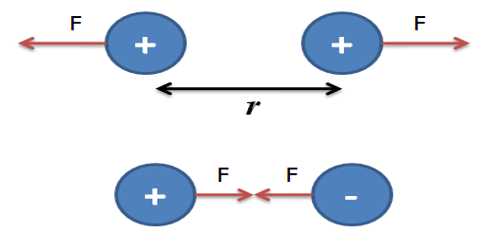
Fig.2 Thus each Na+ ion is surrounded by 6 Cl ions and vice versa. As there are 3 structures, the bond order of 6/3 or 2.Ī bond is not static but dynamic with the atoms undergoing the attractive and repulsive forces as described in the potential well (fig. A coulomb is a measure of electrical charge and is defined as the charge that flows with a constant current of one ampere (1 amp) during one second. The structure of NaCl is two interpenetrating fcc lattices of Na+ and Cl ions as shown in Fig.2. The CO bond order is two, as across the three resonance structures each bond is either 2, 3 or 1, for a total of 6. Express your answer in coulombs to three significant. What is the charge q1 on the particle Ignore the effects of gravity. \) atom.\): Resonance structures for carbon dioxide. Study with Quizlet and memorize flashcards containing terms like Suppose that a charged particle of diameter 1.00 micrometer moves with constant speed in an electric field of magnitude 1.00×105 newtons per coulomb while acted upon by a drag force of 7.25×1011 newtons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
